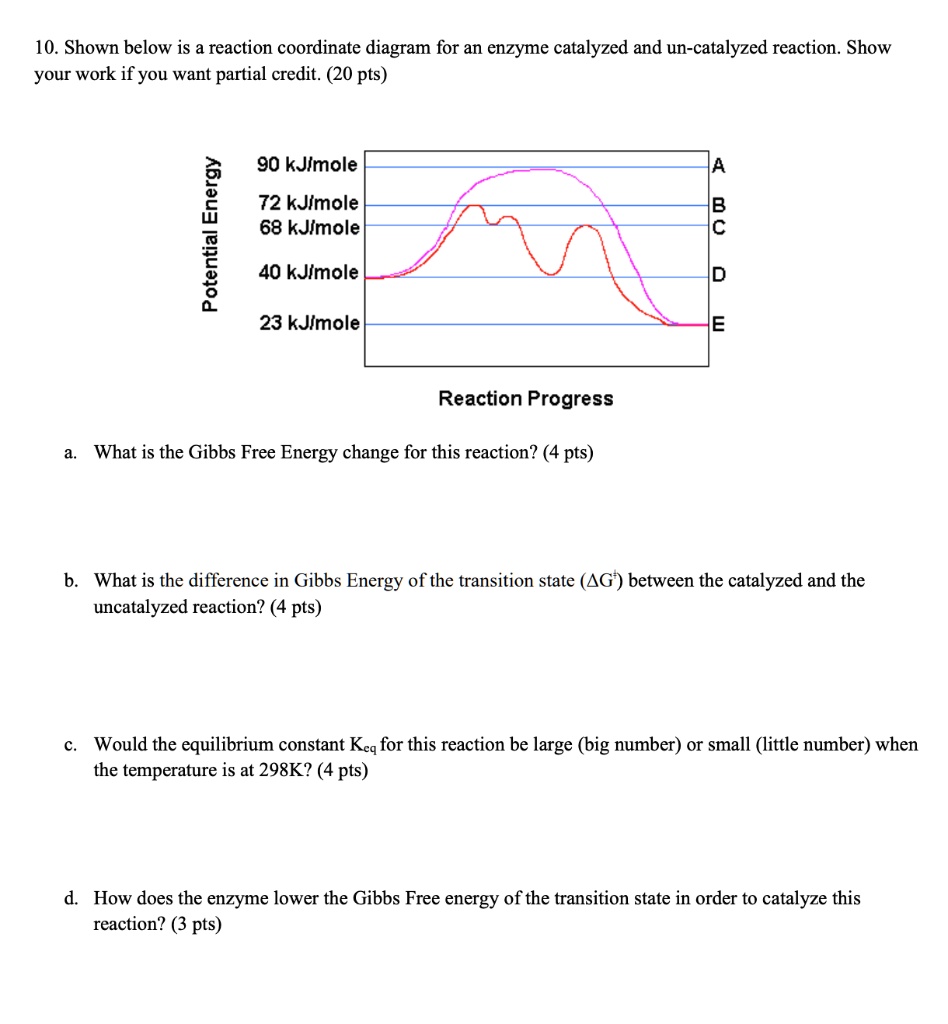
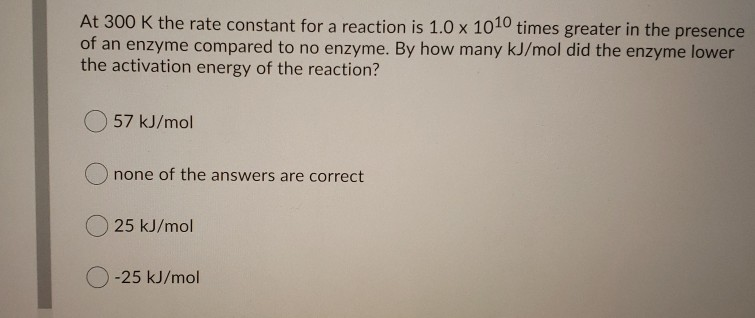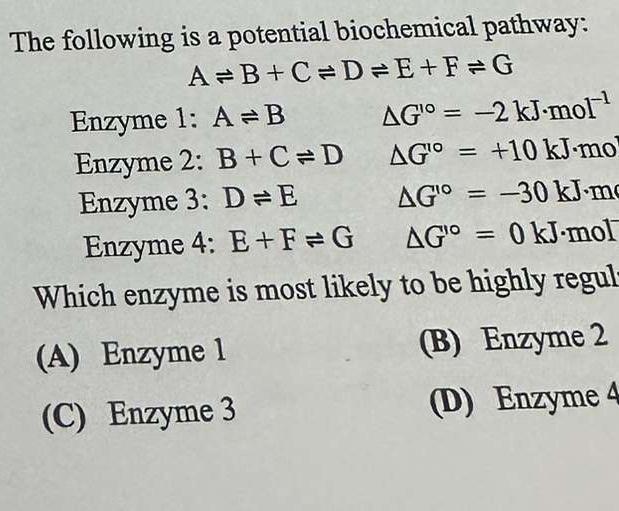The influence of pH on the rates of enzyme reactions. Part 5.—The case of several enzyme-substrate intermediates - Transactions of the Faraday Society (RSC Publishing)

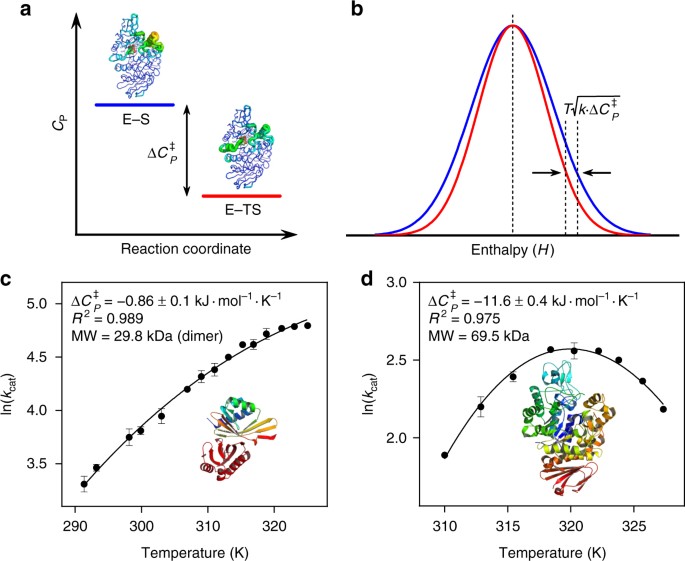
a) Activation energy ( Ea , kJ mol À 1 ), (b) Optimum temperature ( T... | Download Scientific Diagram