
Mild palladium-catalyzed cyanation of (hetero)aryl halides and triflates in aqueous media. - Abstract - Europe PMC

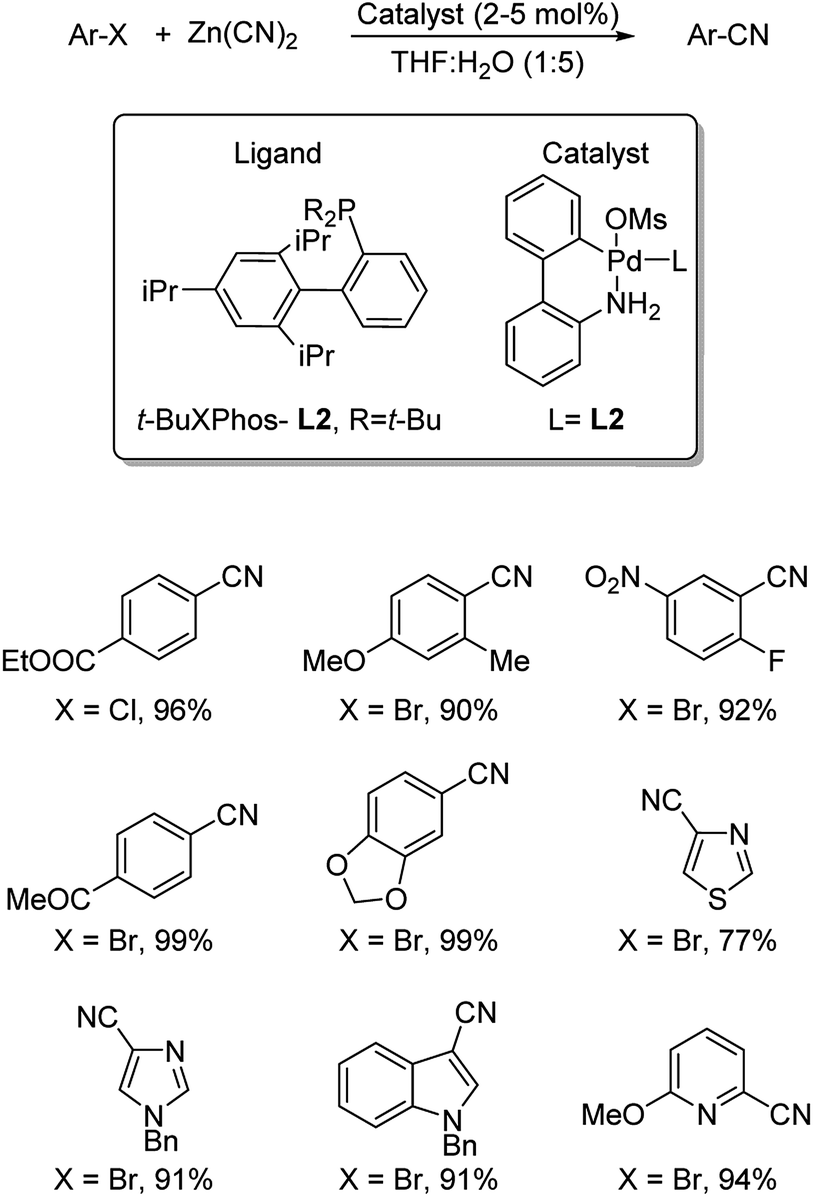
Mild Palladium-Catalyzed Cyanation of (Hetero)aryl Halides and Triflates in Aqueous Media | Organic Letters

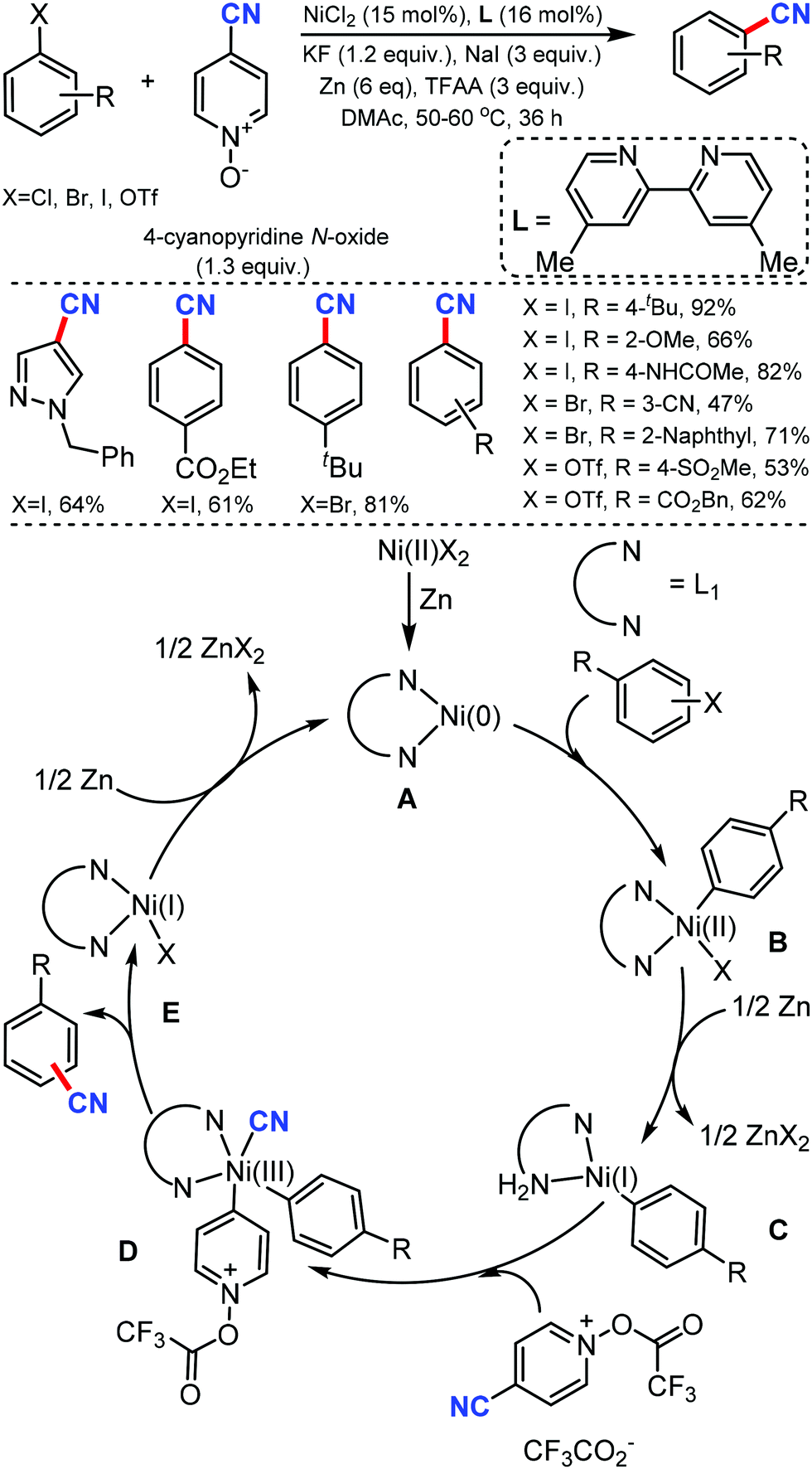
General and Mild Nickel-Catalyzed Cyanation of Aryl/Heteroaryl Chlorides with Zn(CN)2: Key Roles of DMAP | Organic Letters
Recent advances and prospects in the palladium-catalyzed cyanation of aryl halides - RSC Advances (RSC Publishing) DOI:10.1039/D0RA05960A

Nickel-Catalyzed Cyanation of Phenol Derivatives with Zn(CN)2 Involving C–O Bond Cleavage | The Journal of Organic Chemistry

General and Mild Nickel-Catalyzed Cyanation of Aryl/Heteroaryl Chlorides with Zn(CN)2: Key Roles of DMAP | Organic Letters
Recent advances and prospects in the palladium-catalyzed cyanation of aryl halides - RSC Advances (RSC Publishing) DOI:10.1039/D0RA05960A
![PDF] A room temperature cyanation of (hetero)aromatic chlorides by an air stable nickel(ii) XantPhos precatalyst and Zn(CN)2. | Semantic Scholar PDF] A room temperature cyanation of (hetero)aromatic chlorides by an air stable nickel(ii) XantPhos precatalyst and Zn(CN)2. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/20d66ee984041b9b4a0b307a900a1045c6c0111c/2-Table1-1.png)
PDF] A room temperature cyanation of (hetero)aromatic chlorides by an air stable nickel(ii) XantPhos precatalyst and Zn(CN)2. | Semantic Scholar

New trends and applications in cyanation isotope chemistry - Derdau - 2018 - Journal of Labelled Compounds and Radiopharmaceuticals - Wiley Online Library

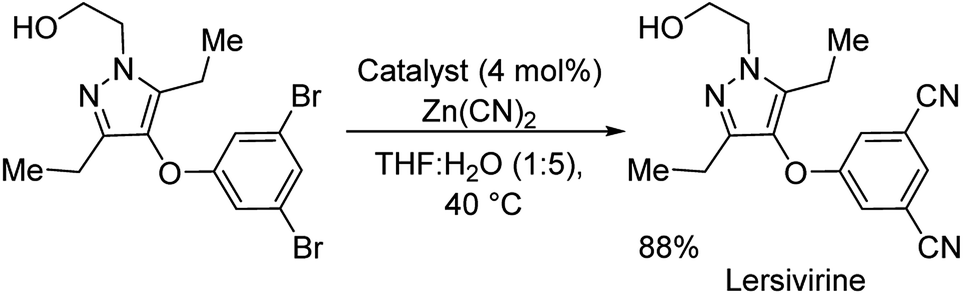
Late‐stage Pd‐catalyzed Cyanations of Aryl/Heteroaryl Halides in Aqueous Micellar Media - Thakore - 2021 - ChemCatChem - Wiley Online Library
C–CN bond formation: an overview of diverse strategies - Chemical Communications (RSC Publishing) DOI:10.1039/D0CC07783F
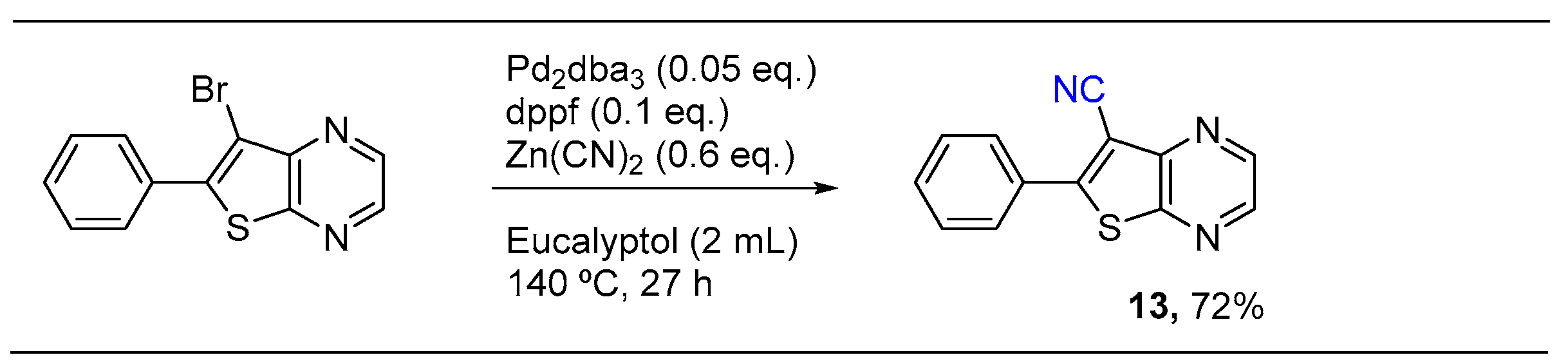
Catalysts | Free Full-Text | Eucalyptol: A Bio-Based Solvent for the Synthesis of O,S,N-Heterocycles. Application to Hiyama Coupling, Cyanation, and Multicomponent Reactions

Applied Sciences | Free Full-Text | Experimental and Process Modelling Investigation of the Hydrogen Generation from Formic Acid Decomposition Using a Pd/Zn Catalyst

General and Mild Nickel-Catalyzed Cyanation of Aryl/Heteroaryl Chlorides with Zn(CN)2: Key Roles of DMAP. | Semantic Scholar
![A mild and efficient palladium-catalyzed cyanation of aryl chlorides with K4[Fe(CN)6]. | Semantic Scholar A mild and efficient palladium-catalyzed cyanation of aryl chlorides with K4[Fe(CN)6]. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/861f9c3deb3dc91f7afcf7091bed27b2ed59c45c/2-Figure1-1.png)
A mild and efficient palladium-catalyzed cyanation of aryl chlorides with K4[Fe(CN)6]. | Semantic Scholar
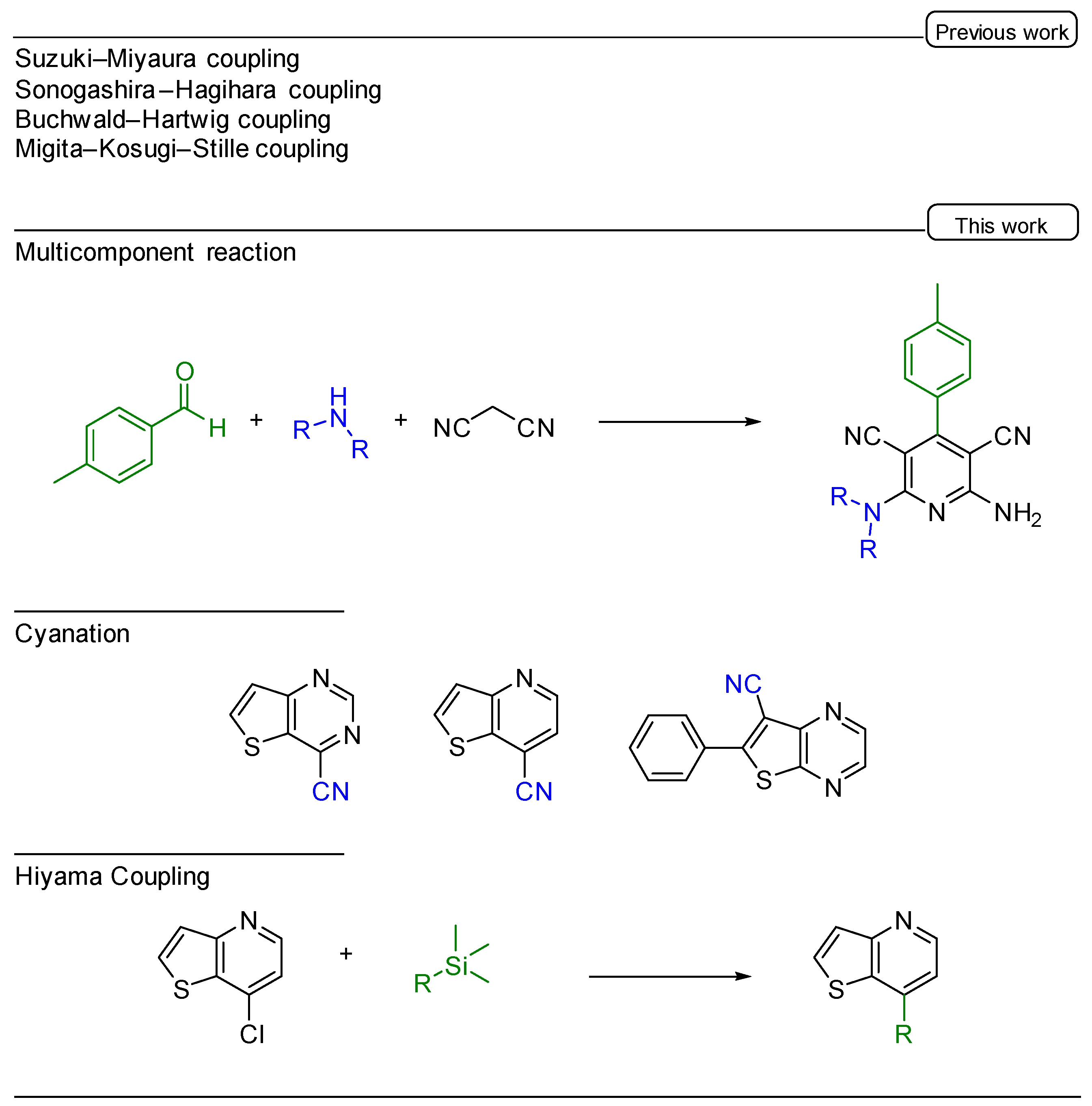
Catalysts | Free Full-Text | Eucalyptol: A Bio-Based Solvent for the Synthesis of O,S,N-Heterocycles. Application to Hiyama Coupling, Cyanation, and Multicomponent Reactions
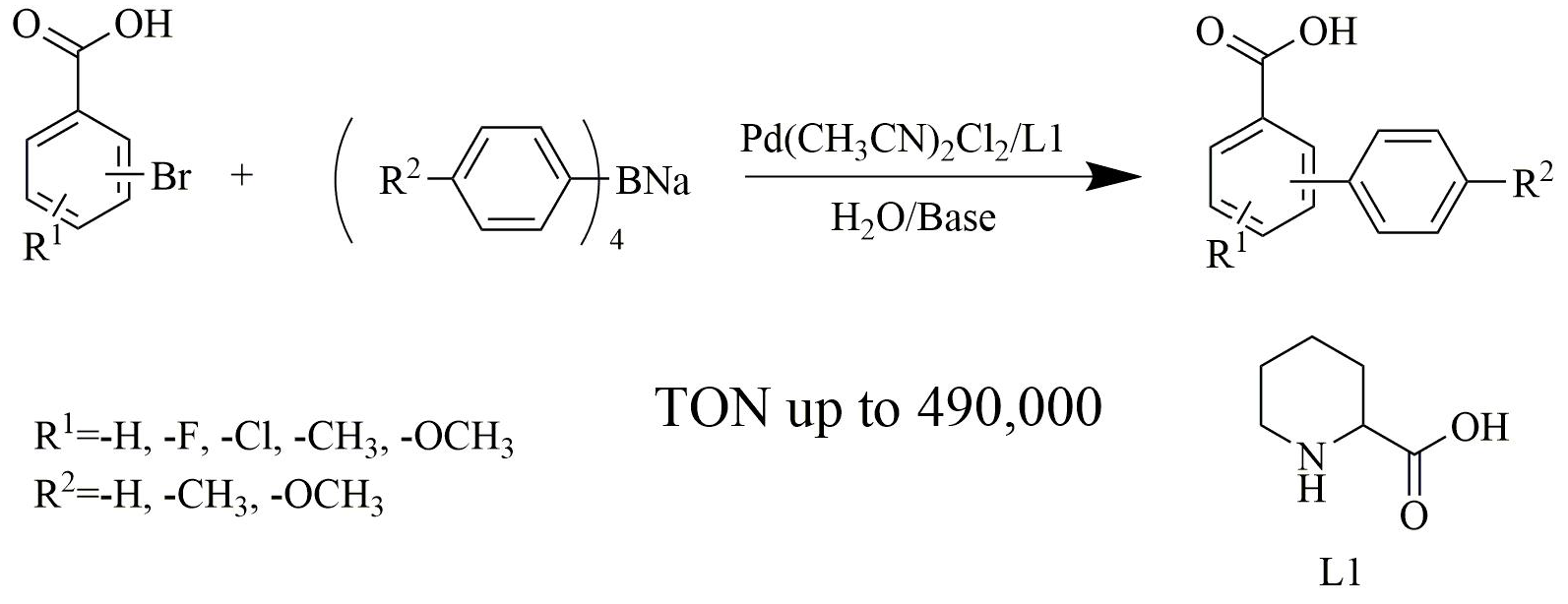
Catalysts | Free Full-Text | Pd(CH3CN)2Cl2/Pipecolinic Acid as a Highly Efficient Catalytic System for Suzuki-Miyaura Cross-coupling Reaction of Bromoaryl Carboxylic Acids in Water




